Article
Feb 11, 2026
Complete guide for participants in bioequivalence clinical studies
Understand how bioequivalence clinical studies work, who can participate, what the benefits are, and how to safely enroll at Synvia.

Clinical bioequivalence studies are part of a crucial step to ensure that generic medications reach the market with the same safety, quality, and effectiveness as the reference (original) medication.
These studies allow millions of people to access reliable treatments at more affordable prices — and the participation of healthy volunteers makes all of this possible.
Synvia is a leader in clinical research in Latin America, with a team of over 800 professionals and one of the largest analytical facilities on the continent.
In 20 years of operation, we have made many contributions to the accuracy and improvement of the medications available in pharmacies across Brazil:
+1,800 studies conducted +30,000 participants 15,000 m² of infrastructure |
These numbers show the scale, responsibility, and seriousness that involve each study conducted by the company. Want to understand more about the subject? Continue reading!
How do clinical bioequivalence studies work?
Clinical bioequivalence studies are conducted with healthy individuals to compare the speed and amount with which the generic medication and the reference (original) medication are absorbed by the body.
It is through these tests that it is proven that both medications behave the same way in the body — ensuring that the generic is as safe and qualified as the original.
These clinical studies also offer participants the opportunity to directly contribute to public health and also receive financial compensation for their time and collaboration.
Do you want to help validate generic medications and receive compensation for your contribution? Click here.

Who can participate in bioequivalence research?
Healthy individuals over the age of 18 who meet the criteria defined for each protocol can participate in clinical bioequivalence studies. The main requirements include:
Be over 18 years old. | Be in good physical condition. | Not use continuous medications, unless approved by a doctor. |
Not have a history of serious illnesses. | Not be pregnant or breastfeeding (in the case of women). | Be willing to fast, collect samples, and adhere to set schedules. |
The confirmation of this eligibility is done during the clinical screening, a stage that ensures safety for all involved.
Click here and be among the first to be called for new bioequivalence studies.
Before the bioequivalence study: how to prepare?
Before participating in clinical bioequivalence studies, it is important to understand that some preparation steps ensure your safety and the quality of the results.
Below, you will find exactly what to do before this moment and how to prepare so that everything goes as smoothly as possible. Let's go?
Preparation for screening
Preparation for screening is simple but essential to ensure that your tests accurately reflect your health status. Small care in the days prior helps avoid changes in results and increases your chances of being approved to participate in the study.
Before proceeding to the evaluation stage, check the guidelines below and make sure everything is in order, so that the study can proceed without difficulties:
Bring an official photo ID: RG or driver's license.
Get a good night's sleep the night before: this helps with test results.
Avoid alcoholic beverages for at least 48 hours before screening.
Avoid intense physical activity: it can alter clinical parameters.
Fast if requested: as per the guidance of the team.
Inform about any recent health issues: transparency is essential.
What happens during clinical screening?
During clinical screening, health professionals perform comprehensive assessments to ensure that the participant is perfectly fit for the advancement of clinical studies.
Medical evaluation: discussion about health, habits, and history.
Laboratory tests: blood and urine to check general parameters.
Vital signs: blood pressure, temperature, heart rate.
Electrocardiogram (when necessary): assessment of heart activity.
Signing the Free and Informed Consent Form (TCLE): document explaining the rights and duties of the participant.
The screening does not guarantee participation — it only confirms whether the person meets the necessary criteria. Register now and receive the invitation as soon as the spots open:
Stages during the study: how each one works
Before detailing each stage, it is important to understand that all clinical bioequivalence studies follow rigorous protocols, approved by Ethics Committees, and are constantly monitored by trained teams.

Do you want to participate in Synvia's bioequivalence research? Click here to access our Whatsapp and register.
Rules and benefits of clinical bioequivalence studies
Clinical bioequivalence studies follow strict guidelines to protect the participant and ensure that all results obtained are truly reliable.
These rules exist to standardize each stage of the study, maintain the safety of everyone and ensure that the data obtained accurately represent the behavior of the medication in the body.
Before participating, it is important to know these guidelines. Below, you will find the main ones.
The rules of clinical bioequivalence studies
DO | DO NOT DO |
✅ Follow fasting and meal times. ✅ Immediately report any discomfort. ✅ Bring requested documents. ✅ Follow the team's guidance. | ❌ Do not consume alcohol before or during the study. ❌ Do not engage in intense exercise in the days leading up. ❌ Do not withhold medical information. ❌ Do not take medications without informing the team. |
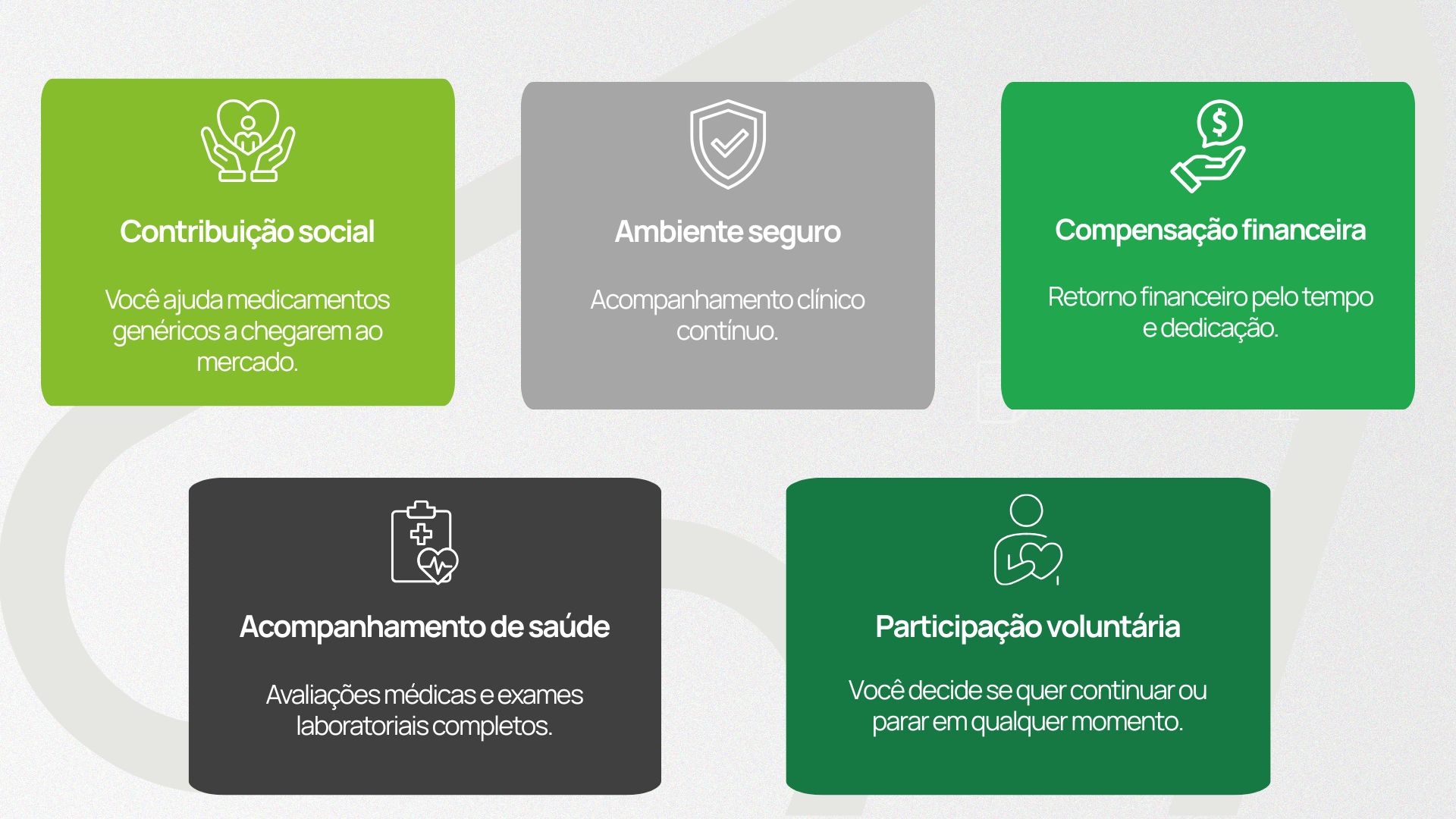
Benefits of participating in bioequivalence studies
Participating in clinical bioequivalence studies is more than contributing to the development of safe and effective generic medications.
The participant also receives direct benefits that make the experience positive, structured, and rewarding:
Ethics, safety, and transparency
The foundation of any clinical study is ethics, and this is especially true in the bioequivalence studies conducted by Synvia.
The entire process is structured to protect the participant, offer complete transparency, and ensure that each stage is monitored by qualified professionals.
Even before starting, the participant receives all necessary information to decide whether they want to participate and is guaranteed that they can withdraw at any time — without justification and without penalty.
Approved by the Research Ethics Committee (CEP).
Constant monitoring by doctors, nurses, and technical staff.
Complete explanation in the TCLE before any procedure.
Right to withdraw at any time, without justification.
How to participate in the next study?
Participating in the next clinical bioequivalence studies is simple and fully guided by Synvia's team. Just fill out your registration by clicking here and wait for the contact for screening.
As new spots open, registrants will be called in order of availability — therefore, the sooner you register, the greater your chance of participating in the next cycle.
If you want to be part of the next studies, directly help in the release of more accessible medications, and also be rewarded for it, just register and follow the next steps.
Your participation is voluntary, totally safe, and extremely important for important research to continue happening in Brazil.
DO YOU WANT TO PARTICIPATE IN THE NEXT STUDY? Click here, register now and receive priority for the next openings. |
Frequently asked questions by participants in bioequivalence research
1. What exactly is a bioequivalence study? |
A bioequivalence study compares a generic medication with its reference version to determine if both have the same behavior in the body — in other words, if the absorption, distribution, and elimination of the active ingredient occur equivalently. This ensures that the generic will have similar efficacy and safety to the original. |
2. Who can participate in these studies? |
Healthy individuals can participate. You must meet the prerequisites below:
|
3. How do I register and get selected? |
You start with a registration — then comes a digital pre-screening to assess eligibility. If your profile matches, a complete check-up (laboratory tests, cardiological exams, etc.) will be scheduled. With tests within the parameters, you will be called to participate in the study. |
4. Do I need to be hospitalized? How long does participation last? |
It depends on the study protocol. Some require hospitalization for collections, monitoring, and administration of the medication; others may have an outpatient component. The time varies based on the study design, but all logistics (hospitalization, tests, meals, rest) are detailed by Synvia's team. |
5. Are the studies safe? Is there a risk for the participant? |
Yes — safety is a priority: all protocols are reviewed and approved by the Research Ethics Committee (CEP), and follow national and international standards (including ANVISA regulation). Participants sign the Free and Informed Consent Form (TCLE) before any procedure, receive clear information about risks, and can withdraw at any time. |
6. What does Synvia offer participants as a benefit? |
Participants have access to free tests and medical monitoring, health follow-up, meals, a comfortable structure during the study, and financial compensation for participation — as a return for the time invested and collaboration with the research. |
7. What if I need to withdraw? Can I leave at any time? |
Yes. Participation is voluntary, and the volunteer can withdraw at any time, without the need to justify and without penalty. This right is guaranteed by research ethics and expressed in the TCLE. |
8. Are health and personal data protected? |
Yes — Synvia follows ethical and regulatory standards that ensure the privacy and confidentiality of participant data. Personal and health information is handled confidentially according to applicable law. |
9. Why are these studies important for Brazil? |
Without bioequivalence studies, generics could not be approved safely. They ensure that generic medications have the same efficacy and safety as reference ones — making treatments more accessible and democratizing access to healthcare in the country. Synvia, with its structure, directly contributes to this process. |
10. How do I know when there are openings to participate in a new study? |
Registration begins with a sign-up (usually via site or form indicated by Synvia). After registration, you enter a database of volunteers. When a new protocol compatible with your profile arises, the team contacts you to start the screening. |